Lesson: Chapter - 5
Solids
As we mentioned above, the molecules that make up solids are generally held
together by ionic or strong covalent bonding, and the attractive forces between
the atoms, ions, or molecules in solids are very strong. In fact, these forces
are so strong that particles in a solid are held in fixed positions and have
very little freedom of movement. Solids have definite shapes and definite
volumes and are not compressible to any extent.
There are a few types of solids
that you should be familiar with for the Chemistry test, and we’ve listed
them below. However, we will start by saying that there are two main categories
of solids—crystalline solids and amorphous solids. Crystalline solids are
those in which the atoms, ions, or molecules that make up the solid exist in a
regular, well-defined arrangement. The smallest repeating pattern of crystalline
solids is known as the unit cell, and unit cells are like bricks in a
wall—they are all identical and repeating. The other main type of solids are
called the amorphous solids. Amorphous solids do not have much order in
their structures. Though their molecules are close together and have little
freedom to move, they are not arranged in a regular order as are those in
crystalline solids. Common examples of this type of solid are glass and
plastics.
There are four types of crystalline solids, all of which you should be familiar with for the exam.
Ionic solids—Made up of positive and negative ions and held together by
electrostatic attractions. They’re characterized by very high melting points and
brittleness and are poor conductors in the solid state. An example of an ionic
solid is table salt, NaCl.
Molecular solids—Made up of atoms or molecules held together by London
dispersion forces, dipole-dipole forces, or hydrogen bonds. Characterized by low
melting points and flexibility and are poor conductors. An example of a
molecular solid is sucrose.
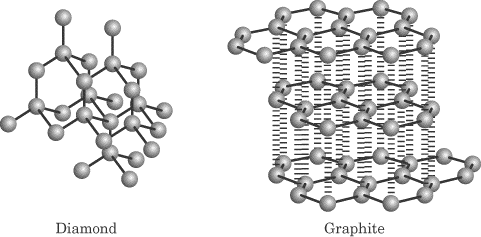
Covalent-network (also called atomic) solids—Made up of atoms connected by
covalent bonds; the intermolecular forces are covalent bonds as well.
Characterized as being very hard with very high melting points and being poor
conductors. Examples of this type of solid are diamond and graphite, and the
fullerenes. As you can see below, graphite has only 2-D hexagonal structure and
therefore is not hard like diamond. The sheets of graphite are held together by
only weak London forces!
Metallic solids—Made up of metal atoms that are held together by metallic
bonds. Characterized by high melting points, can range from soft and malleable
to very hard, and are good conductors of electricity.
Next to display next topic in the chapter.
Practice Questions
Test Prep Lessons With Video Lessons and Explained MCQ
Large number of solved practice MCQ with explanations. Video Lessons and 10 Fully explained Grand/Full Tests.