Lesson: Chapter - 5
Density of Gases
Since gases are mostly empty space, the densities of gases are reported in g/L,
not g/mL as found for solids and liquids. As you’re probably aware,
density is equal to mass per unit of volume. To calculate the density of a gas
at standard temperature and pressure, you take the molecular formula weight of
the gas (grams per mole—from the periodic table) and divide that by the
standard molar volume for a gas, which is 22.4 L per mole:
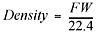
where the formula weight (FW) is in g/mol, and the standard molar volume
is 22.4 L/mol. Now try using this in a problem.
Example
What is the density of helium gas at STP?
Explanation
If the density of the gas is equal to
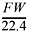
then d = 4.00 g/mol ? 22.4 L/mol, so the density = 0.179 g/L.
If conditions are not standard, we can use this expanded version of the ideal
gas equation:
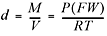
Another really handy rearrangement of the ideal gas equation can be used to find the molecular weight of an
unknown gas.
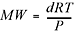
You’ll get a chance to practice
using these in the problems at the end of the chapter. However, there is no need to memorize these last equations
since they are all rearrangements of the ideal gas law. Okay, two more important laws and then we’re finished with
our discussion of gases, and we move on to solutions.
Next to display next topic in the chapter.
Practice Questions
Test Prep Lessons With Video Lessons and Explained MCQ
Large number of solved practice MCQ with explanations. Video Lessons and 10 Fully explained Grand/Full Tests.